PROTAC TECHNOLOGY PLATFORM
PROteolysis TArgeting Chimeras (PROTACs) is an emerging technology in medicinal research. Our team provides related chemistry services to support customers with their targeted protein degredation (TPD) and PROTACs projects. Our highly experienced teams of PROTAC scientists at three of our modern R&D sites located in Boston (USA), Pudong Shanghai (China), and Hengdian Zhejiang (China) can handle the entire process for you with integrated services. From target validation, hit-generation, hit-to-lead, and lead optimization stages, we can support your PROTAC chemistry needs all the way through toxicity and clinical development. With a team of dedicated specialists, modern laboratories, and analytical instrumentation we bring the ability to integrate our PROTAC services at any stage resulting in fully optimized solutions.

PROteolysis TArgeting Chimeras (PROTACs) are very specific compounds that degrade unwanted or harmful proteins in cells. Composed of two heads connected by a linker, PROTACs are bifunctional by design. One end of the PROTAC molecule can bind to specific target proteins while the other end recruits a E3 ubiquitin ligase (cellular enzyme). The PROTAC can then bring the ligase enzyme into close contact with the target protein, which in turn labels the protein with a ubiquitin tag resulting in its degradation by the cell’s ubiquitin-proteasome system. This system is the cell’s natural waste disposal system and will eliminate the undesired protein from the cell. Applications for PROteolysis TArgeting Chimeras (PROTACs) can include slowing down the progression of cancerous tumors or enhancing the targeting ability of small molecule drug therapies.


PROTAC CHEMISTRY SERVICE
We offer chemistry services for PROteolysis TArgeting Chimera (PROTAC) projects, from early-stage drug discovery chemistry to clinical development.
Our skilled PROTAC scientists can assess POI binders, E3 Ligase ligands, exit vectors and linkers, and ternary formations to help determine optimal hit-to-lead approaches for each compound studied. Lead optimization activities may include Absorption, Distribution, Metabolism, and Excretion (ADME) property studies, PK/PD study support, and in-vitro/in-vivo correlation analysis. Apleoa CDMO can use its comprehensive library synthesis approach, based on effective reactions and rational intermediates data, to optimize leads.
All our PROTAC development services are fully supported with complete analytical development, quantification, method validation, and Quality Control (QC) testing services, all performed in our industry leading laboratories by Apeloa’s knowledgeable PROTAC scientists and analysts. Apeloa’s PROTAC analytical services include structure characterization and verification, chiral separations, and development/validation of analytical methods for genotoxic impurities and elemental impurities.

PROTAC INTERMEDIATE COLLECTION
Apeloa’s PROTAC team gained experience and built an intermediate collection by starting with the synthesis of clinical candidate PROTACs. We have currently collected and synthesized over 550 pieces of intermediates, which include various E3 ligase ligands (cereblon, VHL, cIAP, MDM2 ligands, etc), linkers (alkyl, peg, etc), POI binders, E3 ligand-linker conjugates, POI binder-linker conjugates, and reference PROTACs. This extensive collection can facilitate the process of PROTAC library screening and custom synthesis to support the drug hunting effort. Cost-saving and efficient synthesis of chemical moieties can be achieved by our experienced chemistry team.

TARGETED LIBRARY SYNTHESIS PLATFORM
Apeloa offers an integrated library synthesis platform that leads the industry. We apply our unique library generation process to parallel and matrix synthesis approaches. This is to expedite the assessment of fragment combinations and to accelerate the identification of targeted candidates in the most efficient and cost-effective way. All the IP we generate for our customers via the contracted work scope belongs to the customer. This is clearly defined in our balanced CDA/MSA agreements.
Apeloa assesses a wide range of reactions in its synthesis library platform. Some of the PROTAC applied reactions include:
- Click chemistry (N3 – alkyne)
- Reductive amination (NH2-CHO)
- Amide chemistry ((NH2 – carboxylic acid)
- SN2 reactions (Ar-F/alkyl-Br-NH2)
We design our library synthesis by conducting PROTAC scaffold exploration analysis. We look at a wide range of connectors including E3 ligands, linkers, and POI binders from customer, commercial, or custom synthesis sources.
The delivery of our library approach includes batch synthesis of fragments and compounds. Apeloa uses Mass Spectroscopy (MS) to direct the compound purification process.
Apeloa CDMO’s library synthesis platform is ideally suited to support your PROTAC project to an optimized outcome.

PROTAC LINKER OPTIMIZATION
Apeloa CDMO optimizes the linker moiety as this presents opportunities for increased degradation efficiency and enhanced modulation of physico-chemical parameters within the cell of interest. Linker modification is fully supported by Apeloa’s experienced medicinal chemistry team and their use of intermediate collection through the process.
With a range of PEG, Alkyl, and other Gycol linkers available, Apeloa CDMO’s team can provide synthetic accessibility and capability that allows for flexible fine-tuning of linker length. These linker options can be selected based on a variety of parameters including lipophilictity, hydrophilicity, or topological polar surface area (TPSA). Apeloa CDMO’s team also can modify linkers by rigidifying group replacement, such as Alkyne, Piperazine, or Piperidine to provide potency improvements, improved physical properties, and conformational restrictions. Our skilled scientists review the viability of clickable linkers like Triazole, as they can facilitate parallel synthesis and provide potential H-bond interactions in TC.
With Apeloa’s vast array of approaches for linker optimization, we can improve the key parameters such as efficacy, solubility, permeability, and PK profile of the compound. This complex path to optimized drug-like linker moieties can have a large impact on the PROTACs approach for each custom compound.
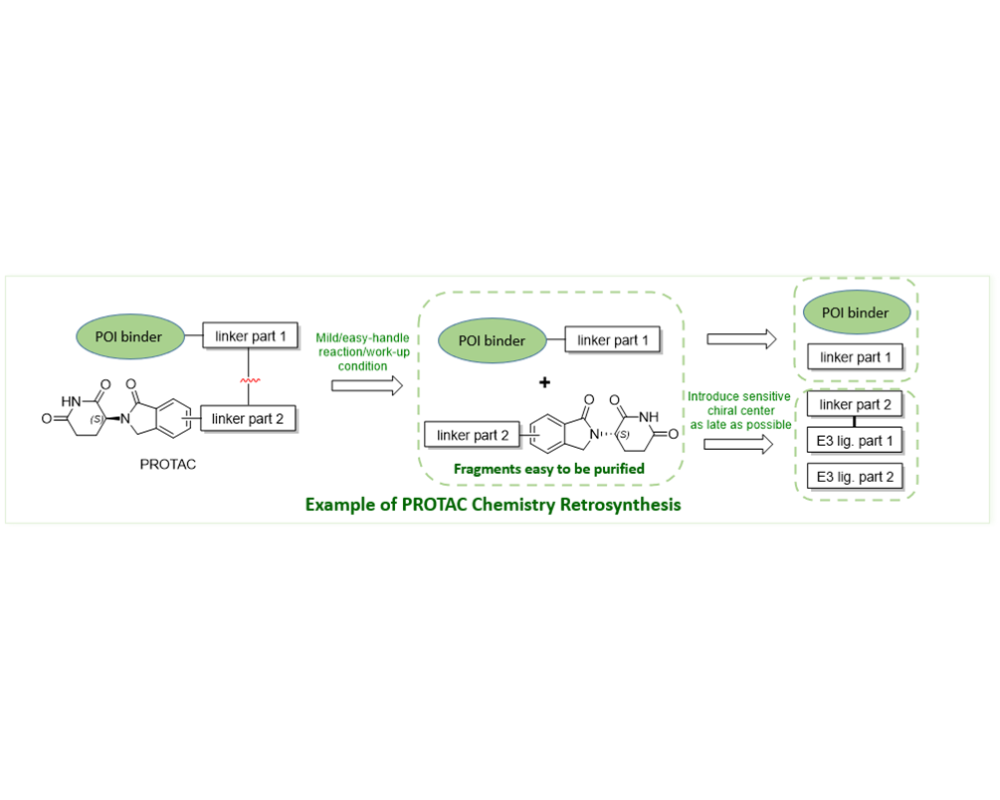
PROTAC SYNTHETIC STRATEGY
Apeloa’s scientific teams can take a retrosynthesis approach in order to avoid epimerization and alleviate purification challenges associated with PROTAC chemistry. Our experienced PROTAC chemists will design a custom fragment assembly process composed of mild, easy-workup, and purification friendly reaction conditions using non-linear synthetic strategy to protect against epimerization. Planning the synthesis to include reactions of fragments with sensitive chiral centers, such as some E3 ligands, can prove effective in the fragment assembly process for PROTACs. It is important to consider a high-standard purification process before the final fragment assembly occurs to ensure optimized conjugation, high yield, and high-quality PROTACs. Apeloa has a strong track record of designing complex but robust fragment purification and assembly processes to create fully optimized PROTACs.
